Amniotic stem cells demonstrate healing potential

Rice University and Texas Children's Hospital scientists are using stem cells from amniotic fluid to promote the growth of robust, functional blood vessels in healing hydrogels.
In new experiments, the lab of bioengineer Jeffrey Jacot combined versatile amniotic stem cells with injectable hydrogels used as scaffolds in regenerative medicine and proved they enhance the development of vessels needed to bring blood to new tissue and carry waste products away.
The results appear in the Journal of Biomedical Materials Research Part A.
Jacot and his colleagues study the use of amniotic fluid cells from pregnant women to help heal infants born with congenital heart defects. Such fluids, drawn during standard tests, are generally discarded but show promise for implants made from a baby's own genetically matched material.
He contends amniotic stem cells are valuable for their ability to differentiate into many other types of cells, including endothelial cells that form blood vessels.
"The main thing we've figured out is how to get a vascularized device: laboratory-grown tissue that is made entirely from amniotic fluid cells," Jacot said. "We showed it's possible to use only cells derived from amniotic fluid."
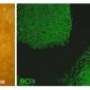
In the lab, researchers from Rice, Texas Children's Hospital and Baylor College of Medicine combined amniotic fluid stem cells with a hydrogel made from polyethylene glycol and fibrin. Fibrin is a biopolymer critical to blood clotting, cellular-matrix interactions, wound healing and angiogenesis, the process by which new vessels branch off from existing ones. Fibrin is widely used as a bioscaffold but suffers from low mechanical stiffness and rapid degradation. Combining fibrin and polyethylene glycol made the hydrogel much more robust, Jacot said.
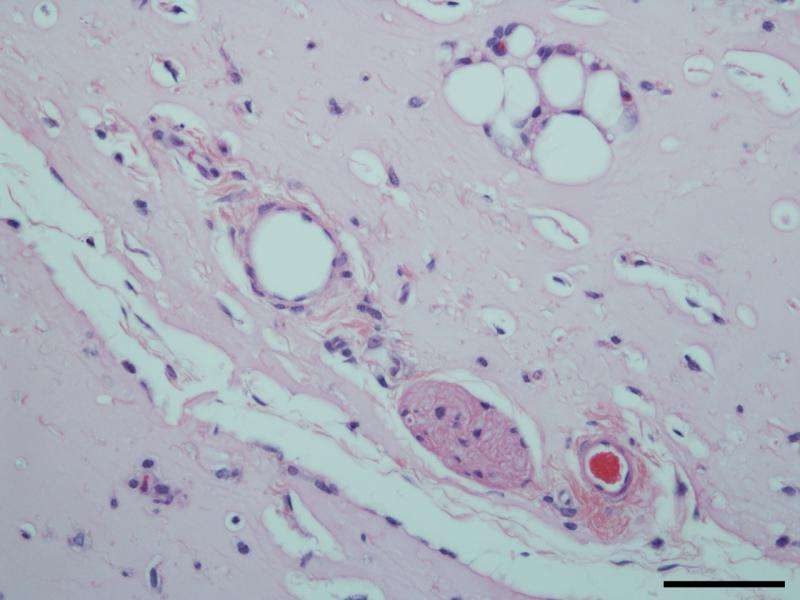
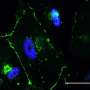
The lab used vascular endothelial growth factor to prompt stem cells to turn into endothelial cells, while the presence of fibrin encouraged the infiltration of native vasculature from neighboring tissue.
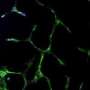
Mice injected with fibrin-only hydrogels showed the development of thin fibril structures, while those infused with the amniotic cell/fibrin hydrogel showed far more robust vasculature, according to the researchers.
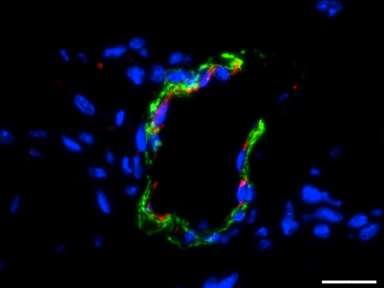
Similar experiments using hydrogel seeded with bone marrow-derived mesenchymal cells also showed vascular growth, but without the guarantee of a tissue match, Jacot said. Seeding with endothelial cells didn't work as well as the researchers expected, he said.
The lab will continue to study the use of amniotic cells to build biocompatible patches for the hearts of infants born with birth defects and for other procedures, Jacot said.
More information: "In situ vascularization of injectable fibrin/poly(ethylene glycol) hydrogels by human amniotic fluid-derived stem cells." Journal of Biomedical Materials Research Part A DOI: 10.1002/jbm.a.35402