Researchers find that immune cells play unexpected role in Lou Gehrig's disease
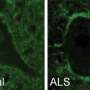
Cedars-Sinai research scientists have found that immune cells in the brain play a direct role in the development of amyotrophic lateral sclerosis, or ALS, offering hope for new therapies to target the neurodegenerative disease that gradually leads to paralysis and death.
The findings will appear in the journal Science on March 18, 2016.
The researchers focused on a genetic mutation that causes ALS, also known as Lou Gehrig's disease, and frontotemporal dementia, another neurological disorder that typically leads to changes in personality, behavior and language.
The investigators developed two genetic strains in mice lacking the gene, known as C9orf72, which they found is important for the function of the immune system in the brain.
Instead of developing ALS, mice without the gene unexpectedly suffered immune system abnormalities. Structures within immune cells—known as lysosomes—that normally dispose of unwanted cellular material stopped functioning properly without the C9orf72 gene.
"The C9orf72 gene is critical for the function of immune cells in the brain, adding to growing evidence that the brain's immune system actively contributes to disease rather than simply responding to injury," said Robert H. Baloh, MD, PhD, senior author of the study and director of Neuromuscular Medicine in the Department of Neurology and the multidisciplinary ALS Program at Cedars-Sinai. "These findings continue a paradigm shift in the way we think of how brain cells are lost in conditions like ALS and Alzheimer's disease."
ALS gradually kills nerve cells in the brain and spinal cord. It is one of the most prevalent neuromuscular diseases, resulting in more than 5,600 new diagnoses in the U.S. each year, according to the ALS Association. Around 10 percent of those diagnoses are caused by the mutation of the C9orf72 gene.
Studies suggest that about 500,000 people in the U.S. are carriers of the mutation.
Baloh and fellow researchers noted that their findings may point the way to new therapies to target immune cell dysfunction, particularly in patients carrying the C9orf72 gene mutation. He said that drugs aimed at decreasing levels of the gene should also be approached with caution because they could further disrupt the immune system.
Jacqueline Gire O'Rourke, PhD, a project scientist who contributed to the study, said the results also could help physicians understand the disparities between carriers of the gene mutation and other ALS patients.
"Our work opens the possibility that C9orf72 gene carriers may even respond differently to immune modulating drugs than other ALS patients," O'Rourke said.
Clive Svendsen, PhD, director of the Board of Governors Regenerative Medicine Institute, where the research was conducted, added that Baloh's research represents an important step toward understanding the role of this particular genetic mutation.
"These new findings will make the field think differently about the causes of ALS," said Svendsen.
More information: "C9orf72 is required for proper macrophage and microglial function in mice" DOI: 10.1126/science.aaf1064