Team reaches milestone in effort to treat bone disorders

A recent study from the University of Nebraska-Lincoln and University of Nebraska Medical Center has reported progress toward the bioengineering of cartilage that could help treat disorders known to disrupt the normal development of bones.
Growth plate disorders alter the length and shape of long bones by affecting the growth of cartilage found at the ends of them. One of the most common genetic growth disorders – the cause of dwarfism – hinders the development of arm and leg bones relative to the head and trunk. Other growth plate disorders, which can also stem from trauma or the treatment of pediatric cancers, affect only specific bones.
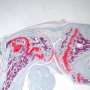
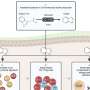
Conventional treatment often involves breaking and resetting a bone, sometimes multiple times, during childhood and adolescence. But researchers from the University of Nebraska-Lincoln and UNMC have documented the first successful effort to establish communication between mature and immature cartilage cells, a necessary step for cartilage and bone growth.
"Our study represents an important step in the engineering of cartilage and is much different than other reports in the tissue engineering field," said Angela Pannier, associate professor of biological systems engineering at the University of Nebraska-Lincoln. "While others have focused on using mature components to build cartilage, we hypothesized that we needed to start with immature cells, and together with the right scaffolding, we could allow the cells to assemble and communicate."
The team reached the milestone by building a growth plate model in the laboratory. By working with mice, the team is seeking answers to detailed questions about how and under what conditions the cells' signaling networks will regulate cartilage growth.
"The hope is that tissue engineering of growth plate cartilage can help reduce the pain, suffering and cost of growth plate disorders," said Andrew Dudley, associate professor of genetics, cell biology and anatomy at UNMC. "Scientists have been able to produce cartilage in the laboratory, but the tissue doesn't grow and isn't mechanically strong. We've been able to stimulate the cells in a way to get very distinct zones of mature and immature cells that talk."
Pannier, Dudley and their colleagues reported their findings in the journal Tissue Engineering Part A.