October 6, 2017 report
Neutrophils found to be helpful, not harmful, after injury
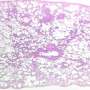
![Dynamic behavior of neutrophils inside sterile injury lesions. (A) Representative images of rapid accumulation of neutrophils in response to focal hepatic injury. Dashed lines highlight injury borders. Scale bars, 230 μm. (B) Representative images of collapsed vessels (arrows) inside the injury area. Scale bar, 10 μm. (C) Quantification of blood vessel diameters inside the injury lesion and in the healthy area (outside injury). Data pooled from three independent experiments. (D) Representative image of neutrophils inside the focal lesion at 12 hours post injury. Higher magnification of the indicated area (white dashed line box) is shown on the right. Scale bar, 30 μm. ****P < 0.0001 [analysis of variance (ANOVA) with Bonferroni’s post hoc test]. Credit: (c) Science (2017). DOI: 10.1126/science.aam9690 Nuetrophils found to be helpful not harmful after injury](https://scx1.b-cdn.net/csz/news/800a/2017/59d75f8a3b784.jpg)
(Medical Xpress)—An international team of researchers has found that neutrophils play an important role in wound cleanup rather than causing unnecessary inflammation. In their paper published in the journal Science, the group describes studying the activities of neutrophils in real time with injured mice. Hannah Garner and Karin de Visser with the Netherlands Cancer Institute offer a Perspective piece on the work done by the team in the same journal issue.
For some time, medical researchers have known that neutrophils offer benefits in responding to injury, but they also believed that neutrophils were detrimental in some patients—immune cells were thought to contribute to unnecessary inflammation, making it more difficult for patients to recover from injuries. As a result, researchers have sought to reduce the actions of neutrophils to speed recovery in traumatized patients. Now, it appears such work may actually have been in vain, as this new effort shows that rather than causing problems, neutrophils actually only help in wound repair.
To learn about the true role of neutrophils as the body reacts to injury, the researchers inflicted burn injuries on test mice and then watched what happened using intravital imaging. They found that rather than causing unnecessary inflammation, the cells initially took apart blood vessels that had collapsed. Several hours later, they were seen picking up damaged DNA fragments. Furthermore, all of the cells moved out and away from the site of the injury within 14 to 16 hours. The team also observed that when they killed the neutrophils, the site of injury contained more debris than sites that with active neutrophils. The researchers continued monitoring the neutrophils and found that a day after an injury had occurred, most of the cells had migrated to bone marrow and were in the process of apoptosis—those few that had not migrated to bone marrow were found in the lungs, but were not causing any problems.
With these new findings, as Garner and de Visser note, more research will likely be conducted regarding the role that neutrophils play in injury repair and suggest that they might lead researchers to new ways of improving treatment of injured patients.
More information: Jing Wang et al. Visualizing the function and fate of neutrophils in sterile injury and repair, Science (2017). DOI: 10.1126/science.aam9690
Abstract
Neutrophils have been implicated as harmful cells in a variety of inappropriate inflammatory conditions where they injure the host, leading to the death of the neutrophils and their subsequent phagocytosis by monocytes and macrophages. Here we show that in a fully repairing sterile thermal hepatic injury, neutrophils also penetrate the injury site and perform the critical tasks of dismantling injured vessels and creating channels for new vascular regrowth. Upon completion of these tasks, they neither die at the injury site nor are phagocytosed. Instead, many of these neutrophils reenter the vasculature and have a preprogrammed journey that entails a sojourn in the lungs to up-regulate CXCR4 (C-X-C motif chemokine receptor 4) before entering the bone marrow, where they undergo apoptosis.
© 2017 Medical Xpress