Team shows how seemingly acute viral infections can persist
Infections caused by viruses, such as respiratory syncytial virus, or RSV, measles, parainfluenza and Ebola, are typically considered acute. These viruses cause disease quickly and live within a host for a limited time. But in some cases the effects of the infection, and presence of the virus itself, can persist. RSV, for example, can lead to chronic respiratory problems, measles can lead to encephalitis and the Ebola virus can be transmitted by patients thought to be cured of the disease.
New findings from the University of Pennsylvania suggest a mechanism that may explain how viruses can linger. Products of viral infection called defective viral genomes, DVGs for short, which have been known to be involved in triggering an immune response, can also kick off a molecular pathway that keeps infected cells alive, the researchers discovered. The study used a novel technique to examine the presence of DVGs on a cell-by-cell basis to show that DVG-enriched cells had strategies to survive in the face of an immune-system attack.
"One of the things the field has known for a long time is that DVGs promote persistent infections in tissue culture," said Carolina B. López, an associate professor of microbiology and immunology in Penn's School of Veterinary Medicine. "But the question was, How do you reconcile that with the fact that they're also very immunostimulatory? How can they help clear virus at the same time as they promote persistence? Our work helps explain this apparent paradox."
López was senior author on the work, teaming with co-lead authors and lab members Jie Xu and Yan Sun. Fellow coauthors included Gordon Ruthel and Daniel Beiting of Penn Vet, Yize Li and Susan R. Weiss of Penn's Perelman School of Medicine and Arjun Raj of the School of Engineering and Applied Science. Their study was published in Nature Communications.
DVGs have been a major focus of López's lab for years. These partial viral genomes are produced in infected cells when a virus begins to replicate rapidly, leading to defective versions of itself that contain large deletions. Once thought not to have any biological function, DVGs are increasingly believed to be important components of viral infections.
In 2013, López and colleagues reported that DVGs were critical in stimulating an immune response to respiratory viruses in mice; when DVGs were depleted from a virus, mice had more severe infections. In 2015 they reported that DVGs are also critical for stimulating an immune response to the human virus RSV, also demonstrating for the first time that the presence of DVGs in human respiratory samples from infected patients correlates with enhanced antiviral immune responses.
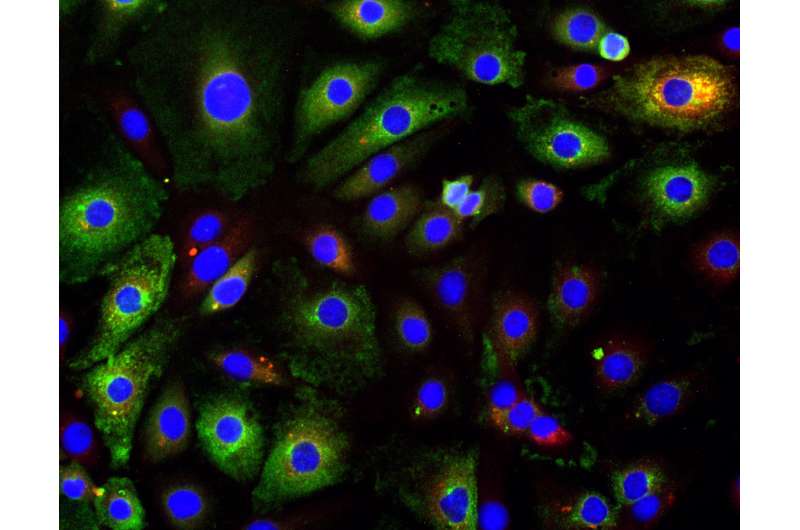
In the current work, López's team used a sophisticated technique that allowed them to differentiate full-length genomes from the partial genomes of DVGs at the single-cell level. They studied cells in culture infected with the Sendai virus, or with RSV, a virus that often affects infants and can lead to chronic respiratory problems,
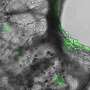
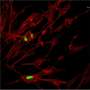
Labeling the full-length genomes in red and the partial DVGs in green, the researchers found differences from cell to cell. Some cells had hardly any DVGs, while others were highly enriched with DVGs, with only a small number of full-length genomes.
"We saw this in many different cell lines and even in infected lungs in mice," López said. "We hadn't appreciated before that there is a lot of heterogeneity in what is going on with these DVGs."
To dig deeper into how the DVGs were influencing the course of infection, the researchers infected cells either with a version of the Sendai virus that lacked DVGs or one enriched in DVGs. The cells infected with the virus high in DVGs survived more than twice as long as those infected with virus lacking DVGs. Adding purified DVGs boosted the cells' survival time, indicating a direct role for the DVGs in promoting cell survival.
The results were similar in parallel experiments with RSV, suggesting that the pro-survival role of DVGs held across viral types.
The researchers next were curious to know what molecular pathways might enable the DVG-rich cells to avoid apoptosis. An analysis of highly-expressed genes in DVG-enriched cells compared to the cells with full-length viral genomes revealed that a host of pro-survival genes were activated in the DVG-rich cells. Notably, these genes encoded signaling proteins of the TNF pathway, known to both boost immunity and cell survival, and IFN, known to play a role in antiviral immunity.
A final set of experiments elucidated the mechanism by which a subset of DVG-enriched cells persisted during viral infection. López and colleagues found that signaling through the proteins MAVS and TNF receptor 2 protects infected cells from apoptosis that is otherwise triggered by TNFα.
"We found this dual role for TNF during these infections," López said. "If TNF binds to a cell that doesn't have the MAVS pathway engaged but is infected, the cell is killed, but, if the cell does have this pathway engaged, then it is protected. MAVS is engaged during the antiviral response, and only cells that have a lot of DVGs activate this pathway. These data show that our cells are wired to survive if they are engaged in an antiviral response, explaining the paradoxical functions of DVGs. It seems that in order to persist, the virus is taking advantage of these host pathways that are there to promote the survival of cells working to eliminate the virus."
The results, though limited to in vitro studies in the current report, point to a way that DVGs could enable "acute" viral infections to linger.
López hopes to build on these findings to be sure they hold in vivo. She's also curious to learn more about the dual roles of TNF, which may help explain why the use of TNF-targeted therapies hasn't always turned out as expected.
"I want to see if there's a way we can harness this pathway to minimize and avoid the persistence of these viruses, which is really relevant if we think about the chronic diseases associated with some of these respiratory viruses," López said.
In addition, she would like to explore how generalizable this pathway is and if it could, perhaps, help explain the problems with viral persistence seen in such infections with the Ebola and Zika viruses.
More information: Jie Xu et al, Replication defective viral genomes exploit a cellular pro-survival mechanism to establish paramyxovirus persistence, Nature Communications (2017). DOI: 10.1038/s41467-017-00909-6