Paving the way to a drug for post-partum depression

For women with postpartum depression—by some estimates, nearly one in five new mothers—the announcement last week of positive results in clinical trials of a drug to help alleviate their symptoms was welcome news.
For Jamie Maguire, an assistant professor of neuroscience at Tufts, it was a moment to savor. That's because the developer of the new drug had built on a discovery Maguire made almost a decade ago, when she was a postdoctoral researcher at UCLA, about specific receptors in the brain that seemed to be implicated in postpartum depression.
At the time, Maguire had made several discoveries "which implicated deficits in neurosteroid signaling in postpartum depression," said Philip Haydon, Annetta and Gustav Grisard Professor of Neuroscience at the Tufts School of Medicine. "Her research demonstrated that there are changes in the expression of receptors in the brain," he said, where neurosteroids are active in the last months of a pregnancy and the first months following delivery.
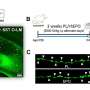
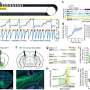
"We saw that animals which lack receptors which are sensitive to these neurosteroids develop maternal depression and exhibit deficits in maternal care," said Maguire. "Based in part on those studies, there was this idea that either there are deficits in neurosteroids themselves or in the actions of neurosteroids that makes this postpartum period vulnerable to mood disorders."
Subsequently, Haydon noted, "she provided the first demonstration that neurosteroids are effective treatments for postpartum depression in this preclinical model." That work, he went on, "paved the way for the future drug discovery efforts for treating postpartum depression."
Sage Therapeutics of Cambridge, Massachusetts, started examining neurosteroids, particularly allopregnanolone, to treat postpartum depression. They created a formulation of that neurosteroid, and on November 9 announced that Phase III clinical trials of the drug, called brexanolone, had been successful, and mitigated postpartum depression symptoms. The firm will now seek FDA approval for the drug. "It is the first treatment specifically targeting this population," said Haydon.
"One of the reasons I'm so excited about this is that there have been so few trials in mental health that have been successful," Maguire said. "There hasn't been a focus on women's health, and particularly postpartum depression—it's a really understudied area."
Maguire, whose work focuses on stress reactivity related to epilepsy as well as postpartum depression, has continued to investigate the mechanisms underlying postpartum depression, building on her earlier discoveries. She and her colleagues have developed another preclinical model that also exhibits postpartum depression-like behaviors, and have been doing basic research work in collaboration with Sage, using the firm's compounds in their models.
Seeing her early work come to fruition is very rewarding, she added. "When you go into basic research, you hope that your work will make an impact, but you accept the fact that you won't likely get to see the impact during your lifetime," Maguire said. "The fact that my research efforts contributed to a successful clinical trial in such a short period of time is so gratifying."