3-D modelling unlocks insight into cancer progression
Medics may soon have a better understanding of how cancer tumours grow and progress, thanks to research from an international collaboration.
The study examined how the various cells that comprise tumours interact, in what is known as the tumour microenvironment (TME). It is published today in the journal Biofabrication.
Senior author Professor David Mooney, from Harvard University, USA, explained: "The TME is characterised by an intricate network of interactions between different cells and the extracellular matrix (ECM). Together, these elements contribute to the microenvironmental regulation of tumour progression and the cancer's spread to other parts of the body.
"Our aim was to better understand how these interactions work, which will ultimately help with predicting and halting the growth and spread of cancer tumours."
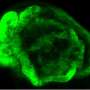
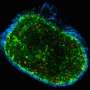
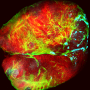
To do this, the researchers developed an in vitro 3-D cellular model of a lung cancer tumour on an interpenetrating hydrogel.
This allowed them to examine how macrophages – a type of cell in the body capable of engulfing and absorbing bacteria and other small cells and particles – and the stiffness of the extracellular matrix influence epithelial-to-mesenchymal transition, which is a fundamental step in cancer cell metastasis.
First author Dr. Marta Alonso-Nocelo, from the Health Research Institute of Santiago de Compostela, Spain, said: "The ECM is not just a supportive structure. It is now recognised as an essential dynamic component, hosting multiple biochemical and mechanical signals that modulate tumour progression.
"Our results showed that, in the absence of the macrophages, changes in ECM stiffness enhanced the spread and invasiveness of the lung cancer cells. When macrophages were present, modified tumour cell growth only occurred when the ECM was in a state of high stiffness."
The study highlights the importance of both mechanical and soluble cues in tumour progression. It also demonstrates how the mechanical properties of the TME can affect tumour cell growth and EMT, as well as impacting how tumour cells regulate and are affected by macrophages.
Professor Mooney said: "These results deepen our understanding of how tumours grow and spread, as well as some of the catalysts for this. More study is needed, but this is a promising step towards finding ways to slow these processes."
More information: Marta Alonso-Nocelo et al. Matrix stiffness and tumor-associated macrophages modulate epithelial to mesenchymal transition of human adenocarcinoma cells, Biofabrication (2018). DOI: 10.1088/1758-5090/aaafbc