Researchers find new trigger for onset of colon cancer, which may lead to better therapies
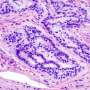
Colon cancer is the second most common cause of cancer-related deaths. The APC protein has long been known for its critical role in preventing colorectal cancer. When APC is inactivated, the development of colorectal cancer is triggered. Inactivation of APC is responsible for the vast majority (80%) of all colorectal cancers. Researchers from the laboratory of Yashi Ahmed, MD, PhD at Dartmouth's Norris Cotton Cancer Center, in collaboration with the groups of Ethan Lee, MD, PhD at Vanderbilt University and David Robbins, PhD at the University of Miami's Sylvester Comprehensive Cancer Center, have identified a new function for this colon cancer gene: APC stops several colon cancer activators.
APC works in a pathway that allows one cell to communicate with its neighbors. When APC is inactivated, this pathway goes into overdrive and that triggers colon cancer development. Exactly how APC acts in this pathway has remained a mystery. The long-held view was that the sole function of APC is to cause the destruction of one activator of the overdrive activity. "The surprise from our research is that APC actually has a second role in putting the brakes on several other activators in the pathway," says Ahmed. "This work changes our view of how this key gene acts, revealing that APC's role is much broader and multi-faceted."
The team's work, "APC Inhibits Ligand-Independent Wnt Signaling by the Clathrin Endocytic Pathway" has been published as a feature article in Developmental Cell.
This new finding about the way that APC blocks the development of colorectal cancer may lead to new therapeutic targets to combat this disease. "Because this new role of APC involves proteins on the cell surface, targeting colorectal cancers may become easier," says Ahmed. "For example, therapeutic antibodies, which normally cannot work inside the cell, can now be used to treat colorectal cancers that have APC mutations."
Figuring out the exact way in which APC stops colon cancer activator proteins will hopefully allow researchers in the future to identify additional drug targets, and to better design therapies for colon cancer patients that kill cancer cells but spare the normal cells in the colon. "The discovery of the new role of APC may also help us understand why APC mutations are so prevalent in certain cancers but not others," says Lee. "Certain tissues may have a backup mechanism to put the brakes on the pathway when APC is mutated."
More information: Kenyi Saito-Diaz et al, APC Inhibits Ligand-Independent Wnt Signaling by the Clathrin Endocytic Pathway, Developmental Cell (2018). DOI: 10.1016/j.devcel.2018.02.013