Some blood cells have a surprising source—your gut
The human intestine may provide up to 10 percent of blood cells in circulation from its own reservoir of blood-forming stem cells, a surprising new study from researchers at Columbia University Vagelos College of Physicians and Surgeons has found.
Scientists had previously thought that blood cells are created exclusively in the bone marrow from a special population of hematopoietic stem cells.
Why It Matters
Intestinal transplantation is the only long-term option for patients with Crohn's and other diseases if their intestines fail. But high rejection rates and life-threatening complications from immunosuppression have limited the success of human intestinal transplantation.
When a person receives a transplanted organ, the immune system often recognizes the new organ as foreign and destroys it. Powerful immunosuppressive drugs blunt these responses, but that makes the patient much more susceptible to infections and other complications.
How Do Blood Cells From the Donor Help the Transplant Recipient?
Analysis of circulating white blood cells in patients after intestinal transplantation suggests that the cells derived from the donated intestine have matured and been educated in the recipient to be tolerant of the recipient's own tissues. Likewise, white blood cells made by the recipient after the transplant may be educated to be tolerant of the donated tissue.
"We are clearly showing that there's immunological cross-talk between the two sets of blood cells that protects the transplant from the patient's immune system and protects the patient from the transplant," says Sykes.
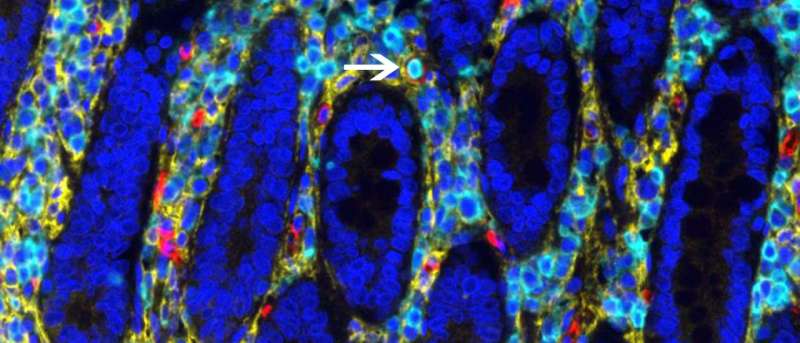
The hematopoietic stem cells in the intestine are eventually replaced by a circulating pool from the recipient, the researchers also found.
How the Finding Could Improve Transplantation
Because patients with more donor blood cells had lower organ rejection rates, the results point toward new strategies for managing organ transplantation.
The intestine's reservoir of blood-forming stem cells was discovered when researchers—led by Megan Sykes, MD, director of the Columbia Center for Translational Immunology—noticed that the blood of patients who had received intestinal transplants contained cells from the donor. The researchers tracked the donor's blood cells back to their source: hematopoietic stem cells in the donated intestine.
The blood cells created from cells in the donor's intestine also may be beneficial to the transplant recipient. The more donor blood cells a patient had in circulation, the less likely they were to reject their transplants.
"It's possible that patients with a high level of donor cells may not require as much immunosuppression as they are currently getting," says Sykes, "and reducing immunosuppression could improve outcomes."
Seeding transplanted organs with additional hematopoietic stem cells from the donor may also increase donor-recipient cross-talk and boost tolerance of the transplant.
"That could improve the lives of transplant patients dramatically," Sykes says. "Our ultimate goal is to get immune tolerance, which would allow us to remove immunosuppression altogether and have the graft treated as self by the patient. That's really the Holy Grail."
What's Next
The researchers are planning a study that will try to boost the number of hematopoietic stem cells delivered during intestinal transplantation, hopefully leading to higher levels of donor blood cells in circulation, immune tolerance, and a reduced need for immunosuppressive drugs.
Other types of transplants may benefit from similar interventions, even for organs that don't appear to carry their own reservoirs of hematopoietic stem cells.
Caveats
This study analyzed 21 patients who had received intestinal transplants.
Although the finding of a novel population of hematopoietic stem cells is exciting, it does not yet justify changes in the current standard of care.
The study, "Human Intestinal Allografts Contain Functional Hematopoietic Stem and Progenitor Cells that are Maintained by a Circulating Pool," was published Nov. 29 in Cell Stem Cell.
More information: Jianing Fu et al. Human Intestinal Allografts Contain Functional Hematopoietic Stem and Progenitor Cells that Are Maintained by a Circulating Pool, Cell Stem Cell (2018). DOI: 10.1016/j.stem.2018.11.007